Electrosynthesis
Electrosynthesis is a rapidly growing field in organic chemistry that uses electricity to drive chemical reactions, offering a sustainable alternative to traditional methods. By replacing conventional oxidizing and reducing agents with electrons as traceless reagents, electrosynthesis allows to reduce waste and thus align with the principles of green chemistry and atom economy. This approach enables selective bond formation, such as C-C, C-N, C-O, and C-S bonds. Its versatility makes it particularly appealing for applications in the fine-chemical and pharmaceutical industries, including the late-stage functionalization of complex drug molecules.
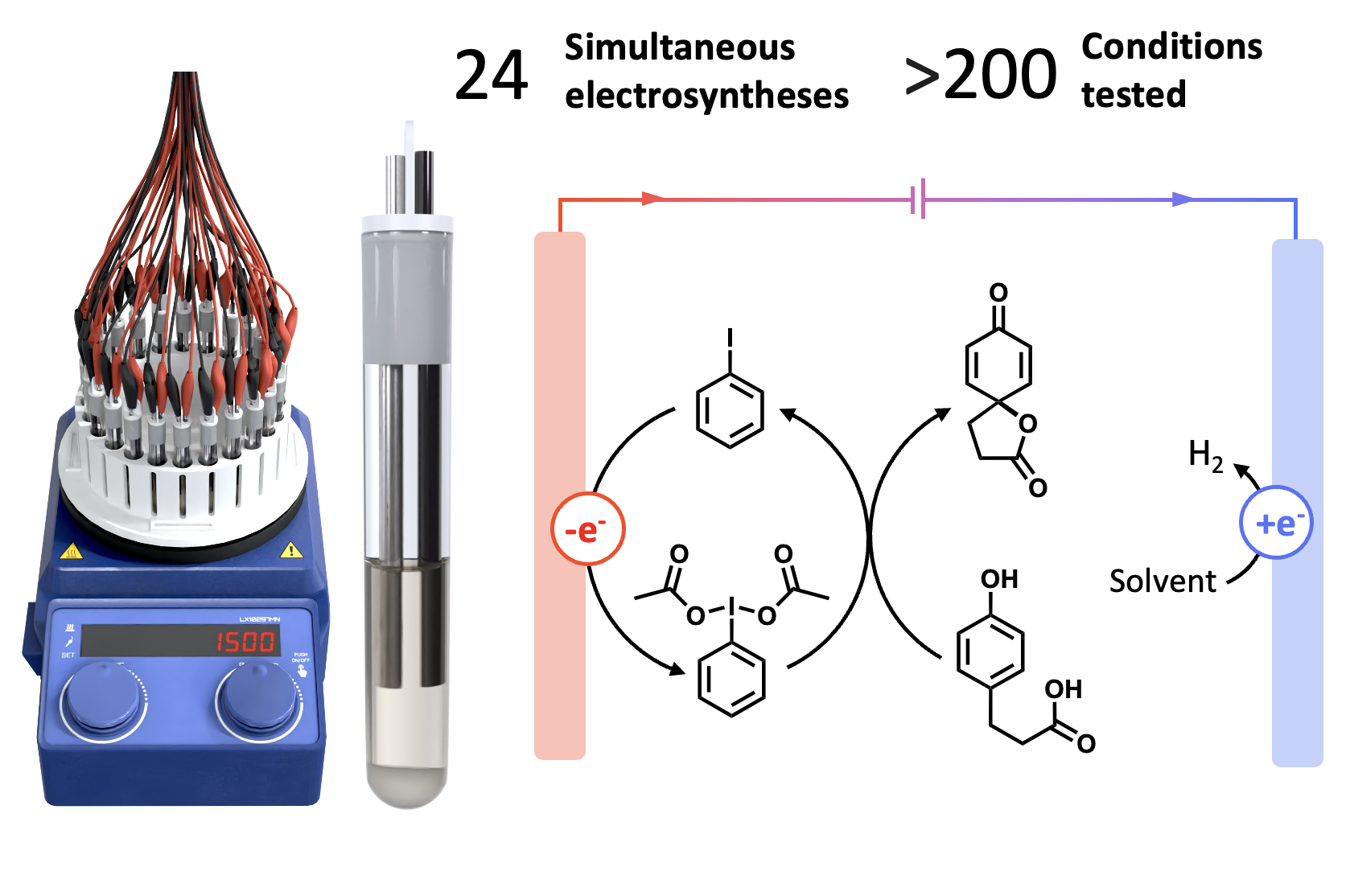
Our lab aims to advance electrosynthetic methodologies by developing innovative tools and exploring diverse reaction types. In collaboration with the McCalla group at McGill University, we designed and built a custom electrochemical reactor capable of running up to 24 simultaneous reactions. This high-throughput device streamlines the optimization of electrochemical parameters, addressing a key bottleneck in electrosynthesis. With access to McGill’s workshop facilities, 3D-printing capabilities, and multiple potentiostats, we have the ability to continuously refine and develop reactors tailored to specific applications.
Our lab aims to advance electrosynthetic methodologies by developing innovative tools and exploring diverse reaction types. In collaboration with the McCalla group at McGill University, we designed and built a custom electrochemical reactor capable of running up to 24 simultaneous reactions. This high-throughput device streamlines the optimization of electrochemical parameters, addressing a key bottleneck in electrosynthesis. With access to McGill’s workshop facilities, 3D-printing capabilities, and multiple potentiostats, we have the ability to continuously refine and develop reactors tailored to specific applications.
Beyond synthesis, we use a wide range of electrochemical and spectroscopic techniques to investigate reaction mechanisms. For example, short-lived, unstable radicals formed electrochemically can be studied using our in-situ electron paramagnetic resonance (EPR) apparatus. In-situ Raman spectroscopy also allows us to detect changes occurring at the electrode surface, while spectroelectrochemistry (UV-Vis) enables us to monitor the formation or decay of species under applied current.
Featured publications
- Juneau, A.; Abdolhosseini, M.; Rocq, C.; Pham, H.; Pascall, M.; Khaliullin, R.; Canesi, S.; McCalla, E.; Mauzeroll, J. Overcoming Challenges in Electrosynthesis Using High-Throughput Electrochemistry: Hypervalent Iodine-Mediated Phenol Dearomatization, a Case Study. ChemElectroChem. 2024, e202400193